Is software a medical device? This is a question that is asked more frequently today than most had expected when the FDA published the Glossary of Computer Systems Software Development Terminology in 1995. The innovation acceleration rate of software development within medical products and services has been beyond even the most optimistic viewpoint of any industry analyst.
To put it in perspective, 1995 was the debut season for the TV show ER, which over its fifteen Golden Globe and Emmy award winning seasons had to make adjustments to the plot and scripting narratives to account for the technological changes within emergency medical centers. If you are not a fan of network TV or pop culture, then you may remember this was the year that IBM officially demoted the suit and put the corporate stamp of approval on “Casual Friday.” Soon after you were replacing your $50 slacks with $100 designer jeans and wondering if America had just invented another holiday to increase retail sales.
The launch of the world wide web in the 1990’s was a significant event in the ever-accelerating rate of technology adoption, enabling the dawn of big data and subsequently 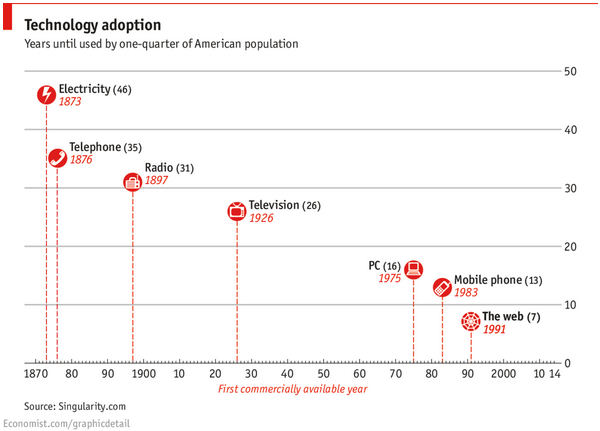 radically altering the manner in which patients researched medical information and communicated with healthcare providers. So much so that Continue reading
radically altering the manner in which patients researched medical information and communicated with healthcare providers. So much so that Continue reading

 (
(